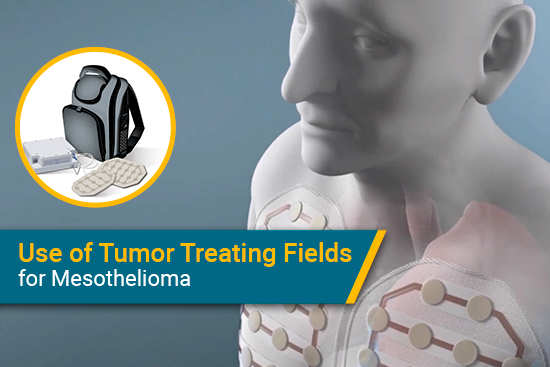
Three years ago, the U.S. Food and Drug Administration made a major announcement: A new therapy was approved as a Humanitarian Use Device for malignant mesothelioma. This treatment, medically known as tumor treating fields, was available for people with mesothelioma who could not have surgery and did not do well with other types of treatment.
After three years of observing patients, doctors and researchers have a real-world understanding of tumor treating fields for mesothelioma – particularly, how often patients wear the device and which side effects they experience.
Doctors at various cancer centers collaborated on a research article published in Cancers. The study tracks five mesothelioma patients through their use of the FDA-approved tumor treating fields device, which is now branded as and called Optune Lua. The analysis reveals how well patients take to wearing the device for the majority of their days.
What Is Tumor Treating Fields Therapy?
Tumor treating fields therapy sends electromagnetic waves from a wearable device through the body. These waves disrupt cell division and prevent cancer cells from replicating. This ultimately can slow the growth of mesothelioma tumors.
The FDA approved a tumor treating fields therapy called NovoTTF-100L System in May 2019. It was the first approval for pleural mesothelioma in 15 years. Patients must receive a chemotherapy combination of pemetrexed and cisplatin while using the device.
The FDA granted it Humanitarian Use Device designation, which is restricted to therapies for rare cancers with fewer than 8,000 cases in the U.S. each year. Mesothelioma affects 2,500-3,000 people each year.
The approval came after NovoTTF-100L led to a median survival time of 18 months for malignant pleural mesothelioma, which forms in the thin lining called the pleura near your lungs. Approximately 60% of patients survived for at least one year, and 97% had a tumor response to the therapy, meaning their cancer either shrunk or stopped growing for an extended amount of time.
As of June 2022, more than 100 U.S. hospitals offer Optune Lua/NovoTTF-100L to patients. It is not the first option for therapy – the approval excludes anyone who can have surgery – but serves as a fallback option if immunotherapy or chemotherapy fail to control the tumors.
Tracking Tumor Treating Fields With Five Patients
Five patients started tumor treating fields therapy. Two patients had stage 1 mesothelioma, two had stage 3 mesothelioma, and one was in stage 2 mesothelioma. All five were not candidates for surgery and tried another type of therapy – chemotherapy, immunotherapy and/or radiation therapy – prior to starting Optune Lua.
All five also received pemetrexed and cisplatin chemotherapy during their tumor treating fields treatment.
Length of Time Wearing the Device Each Day
The wearable device sends electromagnetic waves through the lung cavity throughout the day. Patients may feel a tingling sensation at first but otherwise can go about their usual day without needing to perform any tasks related to the device. The recommended amount of time to wear it is 18 hours a day, per Optune Lua’s manufacturer, Novocure.
The five patients wore the device for an average of 12 hours a day. One of the main symptoms is dermatitis, or skin irritation. The device can cause skin rashes and did so for all five patients. Two patients had grade 1 skin irritation and three had grade 2 skin issues, but no patients suffered from grade 3 dermatitis, so the side effect was mild and controllable. Two patients reduced the time they wore the device due to skin irritation.
One of the areas of growth needed to make the treatment more effective is encouraging and working with patients to wear the device more often during the day. The researchers suggested three schedules to reach the 18-hour marker:
- One 18-hour sprint with six hours of not wearing the device
- Two 9-hour shifts with two three-hour breaks
- Three six-hour shifts with three two-hour breaks
How Long Patients Continued the Treatment
Cycles of tumor treating fields therapy last four weeks (one month). The average number of cycles among the five patients was five. One patient discontinued the treatment after the sixth cycle due to disease progression. Two others discontinued after the fifth and seventh cycles, respectively, due to personal choice.
The other two patients recently started and were still using the treatment at the time the study was published. They were in their second and third cycles of treatment.
Four of the five patients were alive at the follow-up, which was around six months after beginning Optune Lua treatment. Those four have a stable disease, meaning the tumors are not showing signs of growth thanks to the mesothelioma therapy.
Sources & Author
- Feasibility of Tumor Treating Fields with Pemetrexed and Platinum-Based Chemotherapy for Unresectable Malignant Pleural Mesothelioma: Single-Center, Real-World Data. Cancers. Retrieved from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC9032984/. Accessed: 04/27/2022.