
Medically Reviewed By
Karen Ritter, RN BSN
Registered Nurse
JUMP TO A TOPIC
![]()
Important Facts About Immunotherapy for Mesothelioma
- The FDA has approved Keytruda for a small percentage of cases of pleural mesothelioma.
- There are different types of immunotherapy drugs. Most are called “checkpoint inhibitors,” which block cancer cells from evading the immune system.
- Researchers believe immunotherapy can collaborate with other treatments, such as radiation, surgery, and oncolytic viruses.
Immunotherapy Drugs Approved for Mesothelioma
The FDA in 2020 approved Opdivo and Yervoy for mesothelioma. The treatments must be used together and only for cases where surgery isn’t an option. Opdivo and Yervoy are brand names for the checkpoint inhibitor immunotherapy medications nivolumab and ipilimumab, respectively.
Keytruda, another checkpoint inhibitor, is the brand name for the immunotherapy treatment pembrolizumab. The FDA approved it in cases with high amounts of a specific cancerous protein. This approval only works for a small percentage of people with pleural mesothelioma.
How to Get Immunotherapy for Mesothelioma
For patients interested in how to get mesothelioma immunotherapy treatment, here are three steps to receive this promising option:
Look up the side effects and other information about FDA-approved immunotherapy drugs. Be aware of the regular effects so you can know what’s normal and what’s a cause for concern.
Ask your doctor about immunotherapy options offered at the hospital. Opdivo and Yervoy are FDA-approved and available at many cancer centers. Other institutions have clinical trials involving immunotherapy.
Contact a patient advocate for help finding a cancer center. If you don’t already have a mesothelioma specialist, reach out to our team. We can direct you to nearby cancer centers offering immunotherapy treatment.
Makeup of the Immune System
Your immune system has various components. Each plays a role in detecting and responding to mesothelioma and other cancers:
- Antigens – Foreign invaders, such as a virus, that also refer to proteins on cancer cells (tumor antigens)
- T-cells – The soldiers of the immune system, attacking and eliminating cancer cells
- B-cells – Cells that produce antibodies to help defend against antigens
- Antibodies – Proteins that mark cancer cells for the immune system to attack
- Dendritic cells – Messenger cells that download information about cancer cells, including proteins, and present them to T cells
The presence of antigens raises the immune system’s alarm. One of the primary tumor antigens for mesothelioma is a protein called mesothelin.
Mesothelin is the signal for B-cells to produce antibodies. Once they attach to mesothelin, T-cells try to identify and kill the labeled diseased cells.
Immunotherapy can signal to B-cells to produce more antibodies or enhance T-cells against mesothelioma.
How Does Immunotherapy for Mesothelioma Help?
Immunotherapy enables the immune system to properly fight against unwanted intruders, such as cancer cells. Diseases like mesothelioma subdue or sidestep the immune system, allowing it to spread freely without inhibition. Immunotherapy helps the immune system actively target these diseases.
Here’s how some immunotherapy drugs fight cancers like mesothelioma:

Step 1. Immune cells don’t recognize cancerous cells as a threat, which allows them to replicate and spread.

Step 2. Immunotherapy attaches to or kills cancerous cells, which alarms the immune system.

Step 3. The patient’s immune cells recognize cancerous antigens and target cells with similar antigens.
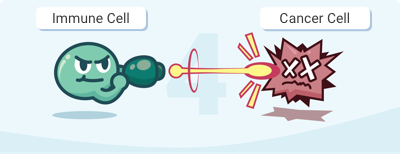
Step 4. The patient’s immune cells seek out the remaining cancerous cells, leaving healthy cells unharmed.
Other immunotherapy drugs add lab-altered T-cells or lab-produced antibodies to aid the immune system.
Fewer Side Effects for Patients
Immunotherapy for mesothelioma has similar side effects to chemotherapy, but they occur less often and less severely. Immunotherapy can make patients fatigued, nauseous or dizzy. Other side effects are weakness, body aches and skin rashes.
Doctors often compare immunotherapy to chemotherapy, hoping to determine which is best for both safety and survival. Most studies favor mesothelioma immunotherapy over chemotherapy.
Link Between Side Effects of Immunotherapy and Survival
Researchers in Japan investigated whether immune-related adverse effects (IRAEs) from the immunotherapy drug nivolumab (Opdivo) led to longer survival. They studied 11 patients, who all received nivolumab (Opdivo) immunotherapy. The results are below:
- The disease control rate was 91% (10 out of 11 patients experienced a stall or shrinkage of their cancer).
- The median overall survival was 15 months.
- Progression-free survival was 6.8 months.
- Eight patients experienced IRAEs, and six had side effects of at least grade 2.
The six patients with grade 2 or higher IRAEs had a progression-free survival of 13 months. The five patients with grade 1 IRAEs or no IRAEs had a progression-free survival of 3.8 months.
The median overall survival was:
- Higher than one year for the group with grade 2 or higher IRAEs
- 8.6 months for the group with grade 1 or no IRAEs
This data shows patients may need to experience discomfort for immunotherapy to be most effective. However, pain and discomfort can be managed with medications and palliative care.
Types of Immunotherapy for Mesothelioma
There are four main types of immunotherapy for mesothelioma: checkpoint inhibitors, oncolytic viruses, adoptive cell therapy and monoclonal antibodies. These types are split into how the drugs stimulate the immune system: actively or passively.
Active immunotherapy uses drugs or viruses to establish a proper immune response. Passive immunotherapy adds laboratory immune cells to aid the body against cancer.
Checkpoint Inhibitors
Checkpoint inhibitors block mesothelioma cells from subduing the immune system T-cells. Blocking the mesothelioma cells allows the immune system to react properly to the presence of mesothelioma. This is a type of active immunotherapy.
Mesothelioma cells have proteins that interact with T-cells’ proteins. The most familiar to doctors and researchers are PD-L1 and PD-1.
PD-1/PD-L1 Checkpoint Inhibitors
PD-L1, the acronym for programmed death-ligand 1, is a surface protein on mesothelioma cells. PD-1, the acronym for programmed cell death protein 1, is a surface protein receptor on T-cells.
When PD-1 binds with PD-L1, the T-cells ignore mesothelioma cells. PD-1/PD-L1 checkpoint inhibitors act as a wall between the protein and receptor. The drugs prevent, or break, the binding and allow T-cells to defend the body.
Examples of PD-1/PD-L1 checkpoint inhibitors include:
- Keytruda, brand name for pembrolizumab
- Opdivo, brand name for nivolumab
- Imfinzi, the brand name for durvalumab
- Tecentriq, brand name for atezolizumab
CTLA-4/B7 Checkpoint Inhibitors
Another example is a CTLA-4/B7 checkpoint inhibitor. These drugs block CTLA-4 (immune cell receptor) and B7 (mesothelioma cell protein).
The main example is Yervoy, the brand name for ipilimumab.
Oncolytic Viruses
Oncolytic viruses deliver an antigen into the body to elicit an immune reaction. This is a combination of oncolytic virus therapy and immunotherapy, and it’s another form of active immunotherapy.
Oncolytic viruses work like regular viruses except they don’t attack healthy cells. They kill only diseased cells, which release antigens that cause an immune response.
An example is mesothelioma oncolytic virus ONCOS-102. The therapy breaks up mesothelioma cells, which releases antigens. The virus then requests for the T-cells to swarm to the site of the cancer.
Adoptive Cell Therapy
Adoptive cell therapy alters immune system cells in a laboratory to better fight cancers like mesothelioma. Usually immunotherapy experts will alter T-cells, which are the soldiers protecting the body from diseases.
The most familiar adoptive cell therapy for mesothelioma is CAR T-cell therapy. CAR is an acronym for chimeric antigen receptor. This form of immunotherapy uses lab-created receptors to target mesothelioma specifically.
IcasM28z is an example of adoptive cell therapy. The CAR T-cells target mesothelin, a protein found on the surface of mesothelioma cells.
Monoclonal Antibodies
Monoclonal antibodies are lab-generated antibodies inserted into the body to fight mesothelioma. It’s another form of passive immunotherapy.
They stick to mesothelioma receptors and partner with the immune system to fight the disease. Examples of monoclonal antibodies are LMB-100, ramucirumab and anetumab ravtansine.
Cancer Vaccines
Cancer vaccines work by introducing a pathogen, such as a bacteria or virus, into the body so the immune system can learn to fight that pathogen. This is accomplished with the help of B cells and T cells, which work together to remember and kill certain pathogens. The pathogen introduced stimulates the immune system to recognize and start killing mesothelioma cells.
How Cancer Vaccines Target Mesothelioma
Cancer vaccines use this targeted approach to stimulate the immune system to attack cancerous cells. The process can be illustrated in four steps.
- Step 1 – Immune cells don’t recognize cancerous cells as a threat, which allows them to replicate and spread.
- Step 2 – The drug is administered to the patient, attaches to cancerous cells and raises the alarm to the immune system.
- Step 3 – The patient’s immune cells are now able to recognize and target cancerous cells on their own.
- Step 4 – The patient’s immune cells seek out and destroy cancerous cells, leaving healthy cells unharmed.
Anti-VEGF (Vascular Endothelial Growth Factor)
Vascular endothelial growth factor (VEGF) is an angiogenic factor produced by many cell types, including malignant tumor cells. VEGF plays a significant role in normal bodily functions, such as bone formation, wound healing and development. For cancer, VEGF stimulates the growth of new blood vessels, which provides nutrients to help tumors grow and multiply.
Anti-VEGF strategies to treat cancers, like mesothelioma, were initially developed to target the pro-angiogenic function of VEGF and prevent the growth of new blood vessels. Preventing the development and growth of new blood vessels deprives cancer cells of oxygen and causes them to suffocate and die.
Combination therapies using anti-VEGF therapies with chemotherapy and/or radiation are effective against many types of tumors due to the targeting of the pro-angiogenic activity and the pro-survival functions of VEGF.
Examples of Anti-VEGF Therapy for Mesothelioma
One example of anti-VEGF therapy for mesothelioma is Apatinib, also known as rivoceranib. This drug is part of the tyrosine kinase inhibitor class of cancer treatment. The inhibitor blocks the VEGF receptor-2.
Apatinib is part of a study for peritoneal mesothelioma. It’s being examined for cases where cytoreductive/HIPEC surgery is not possible.
The cells that make up peritoneal mesothelioma tumors need blood vessels to help them grow strong enough to survive against the immune system. The nutrients also give the peritoneal mesothelioma cells energy to multiply into new cells, continuing the growth and spread of deadly cancer.
Apatinib blocks VEGF by linking with VEGF receptors. Blocking this growth factor puts a brake on angiogenesis, leading to a pause in the creation of additional blood vessels. The effect of this is new cancer cells are not receiving the nutrients they need to survive. The cancer cells eventually die due to a lack of oxygen caused by a lack of nutrients.
In the aforementioned clinical trial, which was published in Frontiers in Oncology, a total of 27 patients received apatinib treatment for peritoneal mesothelioma. The median overall survival was 59.4 months, which is an extraordinary length for any treatment that isn’t surgery.
The overall response rate was nearly 60%. This means the treatment worked in stopping the growth and spread of peritoneal mesothelioma tumors, at least initially, for most patients. Four of the study participants had a partial disease response, meaning the cancer shrunk. Close to half of the patients (12 of 27) had a stable disease, meaning the cancer didn’t show any significant signs of growth or shrinking.
Ramucirumab and bevacizumab are two other anti-VEGF therapies for mesothelioma cancer. These two are in testing for pleural mesothelioma.
Dendritic Cell Therapy
Dendritic cell therapy involves adding dendritic cells to the patient’s body, so the cells can present antigens to the immune system’s T cells, allowing T cells to target and kill cancer cells. Dendritic cells are the messenger cells of the immune system. Consider them as the mail delivery workers.
Dendritic cells are required for an effective immune response and among the most potent antigen-presenting immune cells. Mesothelioma tumors have a low T-cell infiltration. T cells often need to be activated by dendritic cells in order to target and kill tumor cells.
Mesothelioma tumors with a low mutational burden cause low counts of tumor-associated antigens, which makes it difficult for dendritic cells to recognize tumors and activate T cells. Because of this, mesothelioma tumors often have an immunosuppressive environment due to the high volume of immunosuppressive cells and low volume of tumor-associated antigens.
Dendritic cell therapy focuses on the maturation of dendritic cells to make more effective dendritic cells and a larger population. This helps the dendritic cell army identify cancer cells as dangerous and begin the T-cell activation process.

Top Mesothelioma Immunotherapy Drugs
The top immunotherapy drugs for mesothelioma are checkpoint inhibitors. Other immunotherapy drugs include oncolytic viruses and adoptive cell therapies and monoclonal antibodies.
The top immunotherapy drugs for mesothelioma are:
- Nivolumab (Opdivo), a PD-1/PD-L1 checkpoint inhibitor (FDA-approved)
- Ipilimumab (Yervoy), a CTLA-4/B7 checkpoint inhibitor (FDA-approved)
- Pembrolizumab (Keytruda), a PD-1/PD-L1 checkpoint inhibitor (limited FDA approval)
- ONCOS-102, an oncolytic virus (close to FDA approval)
- Ramucirumab, monoclonal antibody
- IcasM28z, an adoptive CAR T-cell therapy
- LMB-100, a monoclonal antibody
- Durvalumab (Imfinzi), a PD-1/PD-L1 checkpoint inhibitor
- Anetumab ravtansine, a monoclonal antibody
- Galinpepimut-S, a cancer vaccine
- Atezolizumab (Tecentriq), a PD-1/PD-L1 checkpoint inhibitor
- Bevacizumab (Avastin), an anti-VEGF treatment
Each is either FDA-approved or part of mesothelioma clinical trials.
Personalized Treatment for Mesothelioma: Potential with Immunotherapy
The holy grail of medicine is personalized treatment for each patient. This idea offers the best chance for long-term survival for everyone, since each person isn’t getting the same treatment.
Every person is different. No one is the exact same, due to each person’s unique genetic makeup. For this reason, no two people’s cancers are the exact same either. Even mesothelioma differs slightly from person to person, which is why a personalized approach can help treat the cancer.
Immunotherapy offers a chance to create personalized mesothelioma treatment.
Dr. Ezra Cohen, of the University of California San Diego Medical Center, is the lead investigator in the Phase 1B trial. The study is titled “Personalized Immunotherapy in Adults With Advanced Cancers” and aims to treat each cancer patient’s disease in a specialized way.
Dr. Cohen joined the podcast to explain how he and other scientists are creating a unique vaccine for “literally one person in the entire world.” Then they create one for another patient, and so on. The trial also involves pembrolizumab, which is the organic name for the immunotherapy drug Keytruda. The trial does not focus solely on mesothelioma cancer, but that doesn’t matter.
Dr. Cohen said the study isn’t testing a drug so much as “a methodology” of personalizing treatment for each individual case.
“It’s identifying the neoantigens and creating the vaccine,” he said. “That methodology is identical in every patient we treat. … The end products, the actual sequences going into the vaccine, are individualized and only go into one person on earth. But the process is the same, and the process is what the FDA is looking at.”
Survival Rates for Mesothelioma Immunotherapy
FDA-approved mesothelioma immunotherapy options range in median survival from 18-20 months. Survival variables include the type of immunotherapy, the patient’s cell type and stage, and whether the drug is paired with other treatment options.
Physician’s Weekly published a study comparing different types of mesothelioma treatment. The main data point was how immunotherapy outperformed chemotherapy. The two types of treatment were often combined with surgery, which immunotherapy also shined:
- 18.2 months for patients having immunotherapy
- 11.7 months for patients having chemotherapy
- 22.6 months for patients having surgery with immunotherapy
- 20.7 months for patients having surgery with chemotherapy
The median survival for patients having immunotherapy was nearly 20 months — including surgery cases and non-surgery cases. The average survival for patients not getting immunotherapy — relying mostly on chemotherapy — was 12.3 months.
The immunotherapy group had a 1-year survival rate of 76%. The chemotherapy group had a 1-year survival rate below 15%.
Immunotherapy Before Mesothelioma Surgery
A study at Baylor College of Medicine tested immunotherapy before surgery for pleural mesothelioma. Doctors said the combination of two immune checkpoint inhibitors — durvalumab and tremelimumab — favorably changed the tumor microenvironment to make cancer cells more vulnerable to T-cell attack. There was an increase in the number of T cells in the tumor.
Survival for Types of Mesothelioma Immunotherapy
The survival benefit and safety are the two measurements the FDA looks for in applications for approval. Most importantly, immunotherapy (with or without other treatments) consistently outperforms chemotherapy for mesothelioma:
- Durvalumab plus chemotherapy had a median survival of 20 months, much better than chemotherapy on its own.
- ONCOS-102 with chemotherapy posted a median survival of 20.5, which is seven months better than just chemotherapy.
- Atezolizumab and bevacizumab, a combination of immunotherapy known as AtezoBev, had 80% of patients survive for at least one year and median progression-free survival was 17.6 months.
- LMB-100 had a median survival of 11.9 months. Of the patients who had high PD-L1 levels, 80% experienced a tumor response. One patient had a complete response, meaning the treatment eliminated the disease.
- Opdivo and Yervoy had a median survival of 18.1 months for pleural mesothelioma patients, which was better than chemotherapy (14.1 months).
- Ramucirumab plus chemotherapy had a median overall survival of almost 14 months, almost double the survival of chemotherapy alone (7.5 months).
- Anetumab ravtansine has the best success when 6.5mg/kg of the drug is administered once every three weeks. The overall response rate was 31%, meaning the disease shrank. The disease control rate was 75%, meaning it either shrank or stopped spreading.
- Dendritic cell therapy had a median survival of 27 months.
- Opdivo and Yervoy also have a significant effect on epithelioid and sarcomatoid mesothelioma patients, the toughest cell histology for mesothelioma. A study out of Germany proved the success of the immunotherapy drugs:
- Epithelioid patients survived on average 19 months with immunotherapy – 3 months longer than chemotherapy.
- Sarcomatoid patients survived for 17 months with immunotherapy, which is far better than chemotherapy (9 months).
Immunotherapy in Multimodal Treatment for Mesothelioma
The FDA is considering immunotherapy as an alternative to chemotherapy, specifically for people who cannot have surgery. However, studies continue to investigate whether immunotherapy works as part of multimodal treatment for mesothelioma.
Multimodal treatment refers to using multiple types of therapy. As a multimodal therapy, immunotherapy would pair with one or multiple of:
- Surgery
- Chemotherapy
- Radiation
Ongoing clinical trials are testing immunotherapy before, after, or both before and after mesothelioma surgery. This is especially relevant for patients with a high amount of PD-L1 expression on their cancer cells.
A report published in the Annals of Thoracic Surgery noted a very poor survival for these cases. All 75 cases in the study involved a mesothelioma surgery and PD-L1 seemed to erase the progress of a resection.
Dr. Patrick Forde, of Johns Hopkins Hospital in Baltimore, Maryland, is running a study using Opdivo and Yervoy six weeks before surgery. Patients can resume immunotherapy weeks after surgery if they choose.
Doctors often pair investigative immunotherapy drugs with mesothelioma chemotherapy in clinical studies. The combination of cancer-killing drugs with immune-enhancing drugs attacks mesothelioma on two fronts.
An example is the pairing of ramucirumab, a monoclonal antibody, with the chemotherapy drug gemcitabine. The combination led to median survival of 14 months — and a one-year survival rate of 56% — compared to seven months for just chemotherapy.
Many doctors feel immunotherapy and mesothelioma radiation therapy can have a synergetic relationship. Radiation inflames the tissue where cancer exists, which can elicit an immune response. Immunotherapy helps make the responding T-cells prepared to fight.
Radiation also can partially kill tumors, clumps of diseased cells, which then release antigens. These antigens are what the T-cells look for, and immunotherapy can amplify the immune system’s ability.
“As immunotherapy is coming more in favor, that may make radiation more in favor,” said Dr. Charles Simone, the chief medical officer of the New York Proton Center. “We have more evidence that immunotherapy and radiation together may have a synergistic effect.”
Frequently Asked Questions About Immunotherapy for Mesothelioma
Does immunotherapy work for mesothelioma?
Immunotherapy is one of the emerging treatment options for mesothelioma patients who cannot undergo surgery. The therapy helps the immune system locate mesothelioma cells and attack the tumors, which slows the disease’s progress and extends the patient’s life. Numerous studies report survival times of multiple years thanks to immunotherapy.
What types of immunotherapy are available to mesothelioma patients?
The most popular type of immunotherapy for mesothelioma is a checkpoint inhibitor. This blocks two protein receptors from linking, which strengthens the immune system. Examples of checkpoint inhibitor drugs are Keytruda, Opdivo and Yervoy. Another example of immunotherapy is giving the body a virus, or something similar, to activate the immune system.
Is Keytruda available for mesothelioma?
The FDA recently approved Keytruda for specific cases of pleural mesothelioma. The drug is for patients who have an unresectable, malignant disease and meet a specific threshold of PD-L1 tumor burden. PD-L1 is a cancerous protein that links with PD-1, an immune system receptor, and inhibits the immune system response. Keytruda blocks the connection.
How can I get immunotherapy to treat my mesothelioma?
The FDA recently approved multiple forms of immunotherapy for mesothelioma, making the treatment more easily accessible through cancer centers. The combination of Opdivo and Yervoy is available to patients who cannot undergo surgery. Patients who don’t meet the FDA’s criteria can enroll in clinical trials to receive immunotherapy on an experimental basis.
Sources & Author
- Types of Immunotherapy. WebMD. Retrieved from: https://www.webmd.com/cancer/immunotherapy-treatment-types. Accessed: 02/24/2021.
- Prognostic Role of Programmed Cell Death 1 Ligand 1 (PD-L1) in Resectable Pleural Mesothelioma. Annals of Thoracic Surgery. Retrieved from: https://pubmed.ncbi.nlm.nih.gov/33248997/. Accessed: 12/14/2020.
- Ramucirumab–gemcitabine encouraging in second-line malignant pleural mesothelioma. Medwire News. Retrieved from:
https://www.medwirenews.com/oncology/respiratory/ramucirumab-gemcitabine-malignant-pleural-mesothelioma/18096212. Accessed: 06/24/2020. - Durvalumab added to standard chemotherapy improved overall survival in mesothelioma. American Association for the Advancement of Science. Retrieved from:
https://eurekalert.org/pub_releases/2020-05/ecrg-dat052020.php. Accessed: 05/21/2020. - FDA Approves Drug Combination for Treating Mesothelioma. U.S. Food and Drug Administration. Retrieved from: https://www.fda.gov/news-events/press-announcements/fda-approves-drug-combination-treating-mesothelioma. Accessed: 10/04/2020.
- Nivolumab Plus Ipilimumab Improves OS in Malignant Pleural Mesothelioma. Cancer Network. Retrieved from: https://www.cancernetwork.com/view/nivolumab-plus-ipilimumab-improves-os-in-malignant-pleural-mesothelioma. Accessed: 08/11/2020.
- Continued survival benefit in Targovax’s ONCOS-102 trial in mesothelioma at the 21-month follow-up. PRNewswire. Retrieved from: https://www.prnewswire.com/news-releases/continued-survival-benefit-in-targovaxs-oncos-102-trial-in-mesothelioma-at-the-21-month-follow-up-301233073.html. Accessed: 02/23/2021.
- Multimodal Therapy Survival in Malignant Pleural Mesothelioma Patients. Physician’s Weekly. Retrieved from: https://www.physiciansweekly.com/multimodal-therapy-survival-in-malignant-pleural-mesothelioma-patients. Accessed: 12/12/2022.
- Efficacy and Adverse Events of Apatinib Salvage Treatment for Refractory Diffuse Malignant Peritoneal Mesothelioma: A Pilot Study. Frontiers in Oncology. Retrieved from: https://pubmed.ncbi.nlm.nih.gov/35847956/. Accessed: 09/28/2022.
- Preoperative immunotherapy for mesothelioma shows favorable outcomes. Baylor College of Medicine. Retrieved from: https://www.bcm.edu/news/preoperative-immunotherapy-for-mesothelioma-shows-favorable-outcomes. Accessed: 12/14/2022.













